932
932  150
1501. Technical Challenges in Precious Metal Analysis
Precious metal analysis is one of the most important applications of X-ray fluorescence (XRF) technology.
XRF is widely used to rapidly analyze elemental composition and purity in jewelry testing, precious metal recycling, and refining industries. However, precious metal systems present several unique analytical challenges compared with common metals.
1.1. Very Close Elemental Energies
Many precious metals belong to high-atomic-number elements, whose characteristic X-ray energies are very close to each other.
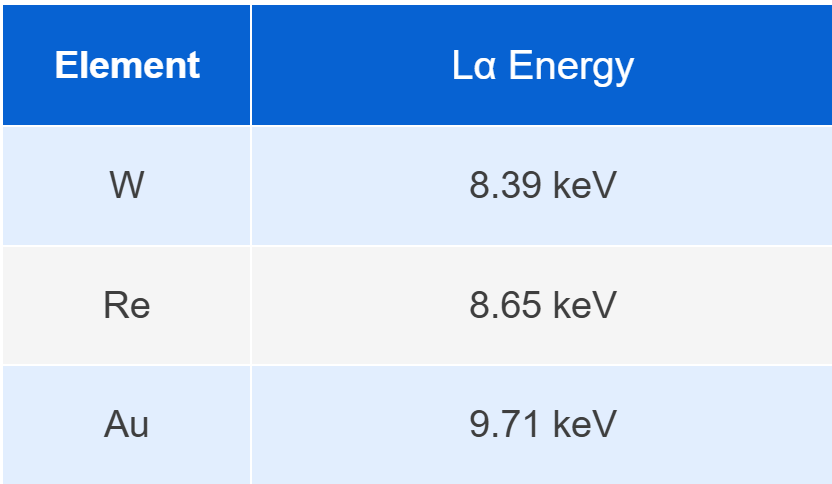
For example:
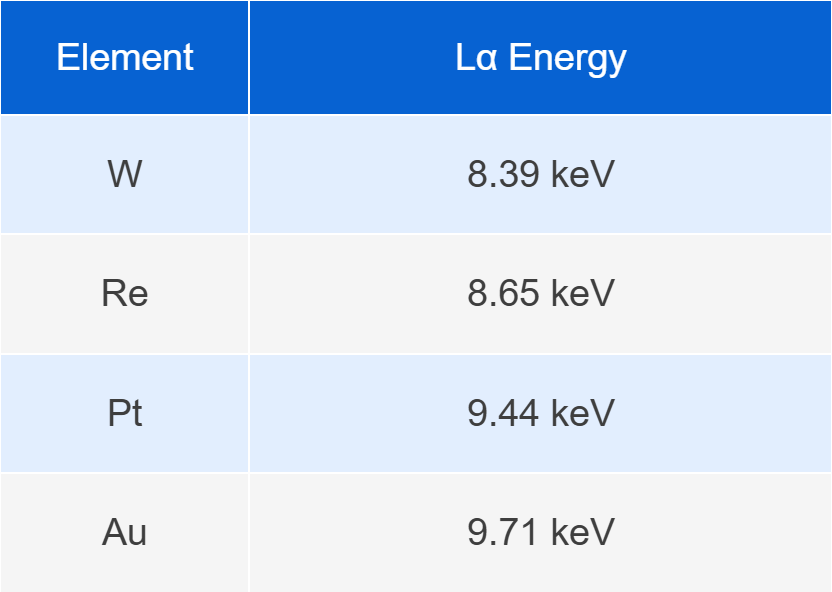
The energy differences between these elements are typically only 200–300 eV. If the detector resolution is insufficient, these spectral peaks may overlap, which can significantly affect element identification and quantitative analysis.
1.2 Complex Alloy Systems
Modern precious metal materials are multi-metal alloys rather than pure elements. Examples include:
• Au–Cu–Ag alloys(K gold)
• Pt–Ir alloys
• Au–Pt–Pd alloys
Taking copper impurities in gold as an example:
The escape peak of Au Lα (Au Lα − E) overlaps almost exactly with Cu Kα peak. When comparing spectra from spectrometers equipped with SIPIN detectors and FSDD detectors:
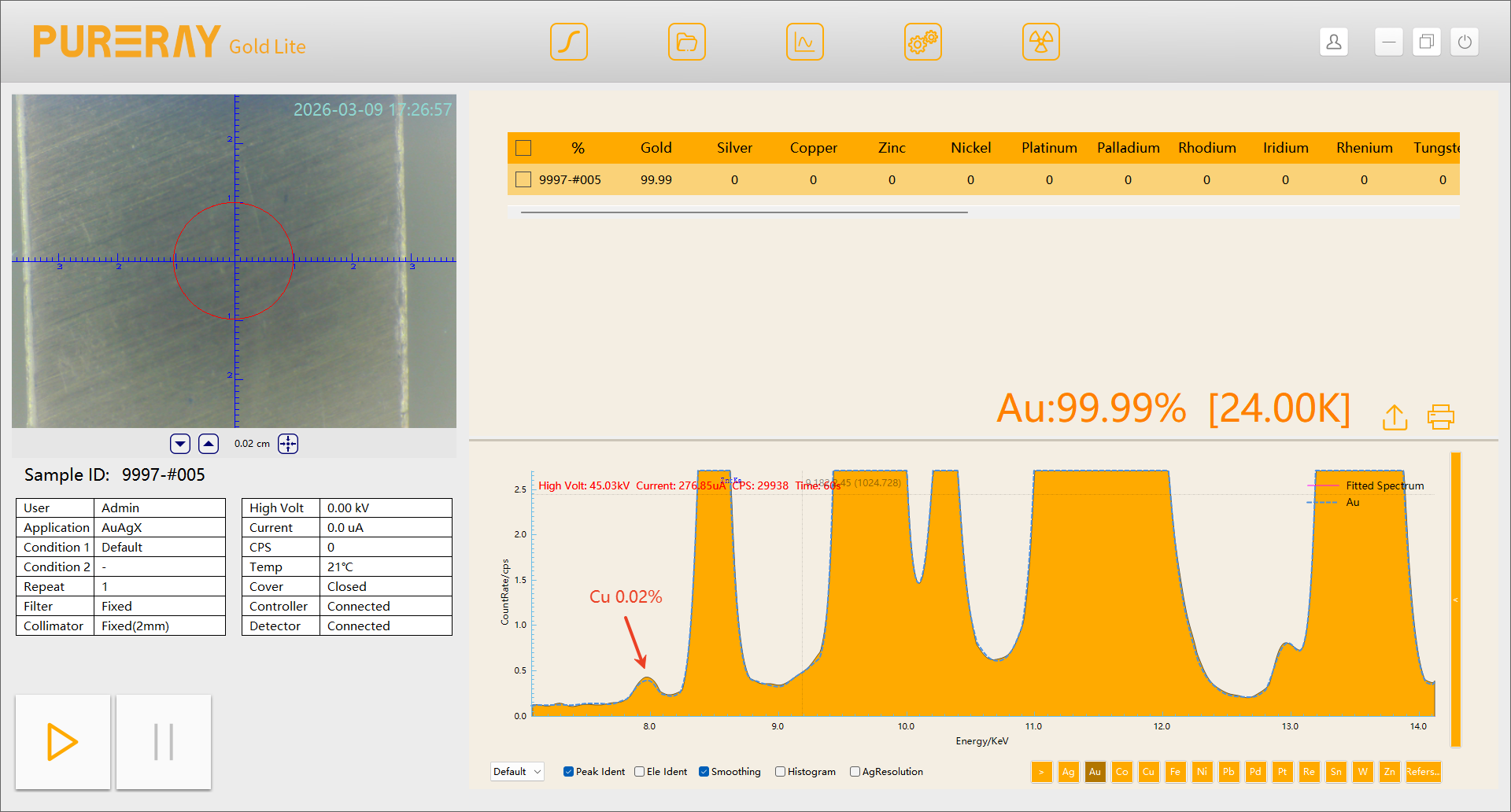
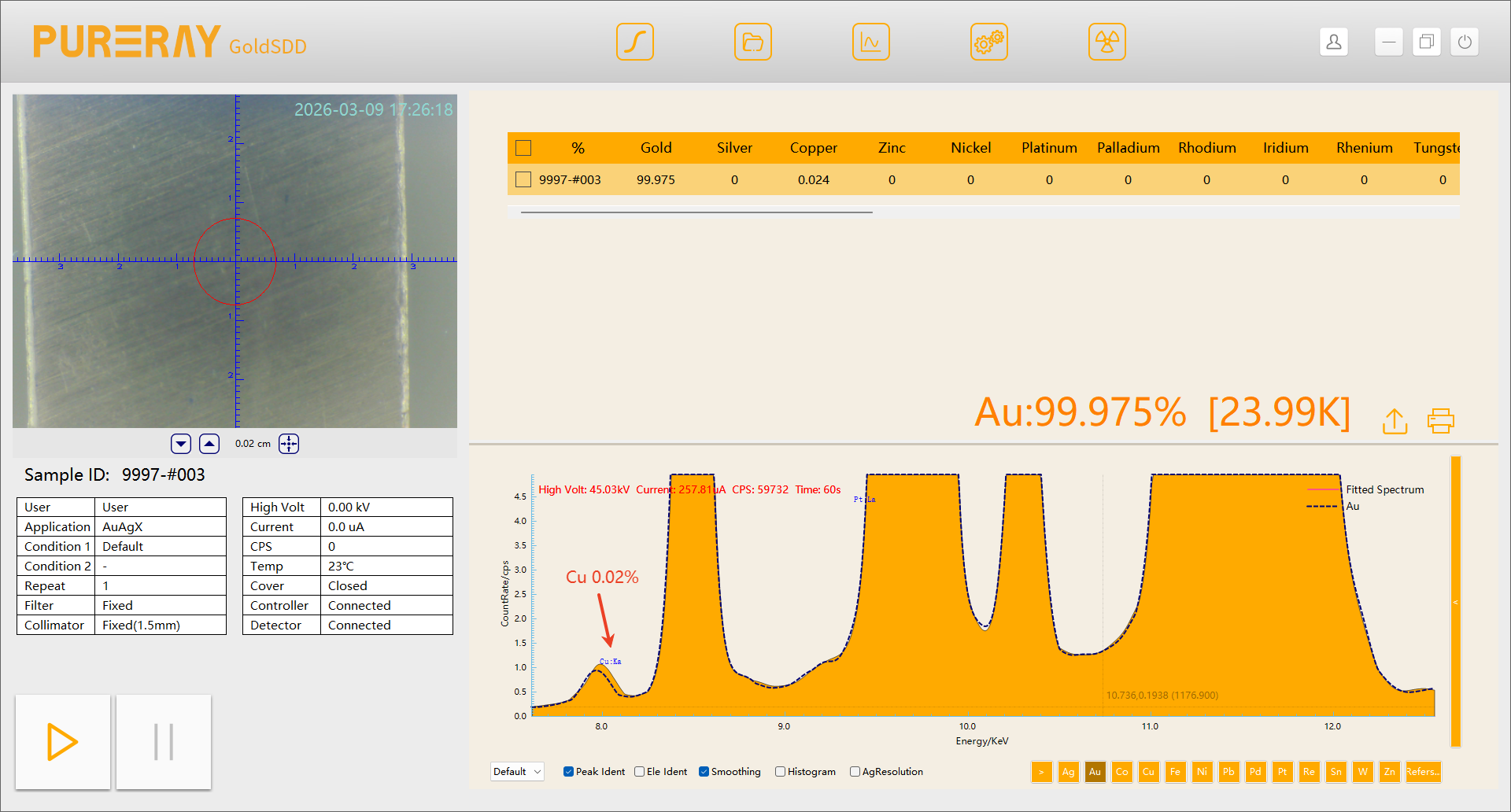
A trace amount of 0.02% Cu cannot be detected by the SiPIN system, whereas the FSDD system clearly resolves the Cu characteristic peak.
1.3. Extremely High Accuracy Requirements
In the jewelry industry, differences in gold content are often only 0.01% – 0.2%. Therefore, analyzes must provide:
• High stability
• High repeatability
• High energy resolution
2. Energy Resolution in Precious Metal Testing
In precious metal analysis, energy resolution directly determines:
• Peak separation capability
• Trace element detection ability
• Quantitative accuracy of alloys
Typical detector resolutions are 125–140 eV for SDD detectors and 165–190 eV for SiPIN detectors.
The narrower spectral peaks provided by SDD detectors enable clearer element identification and more accurate quantitative analysis.
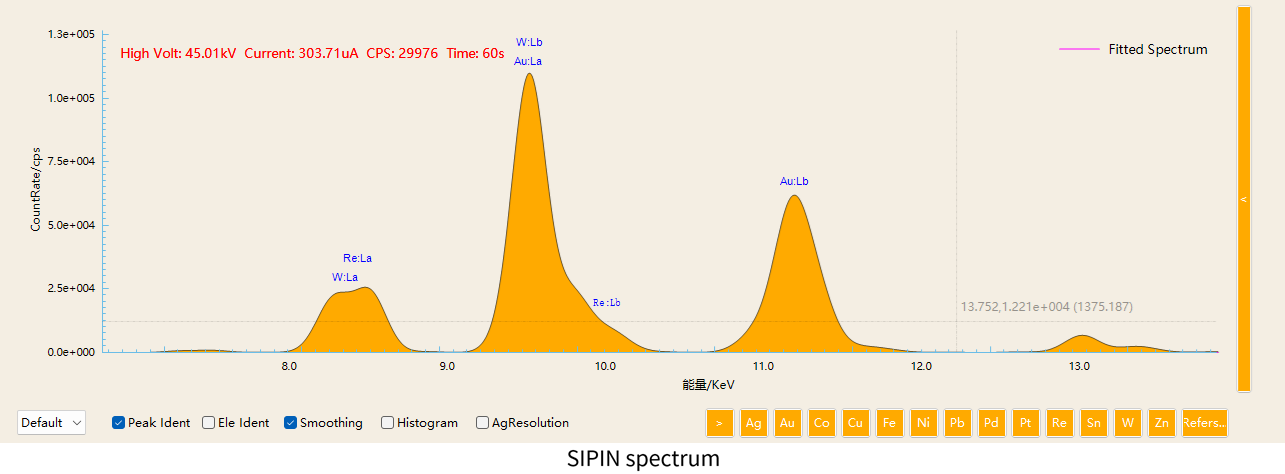
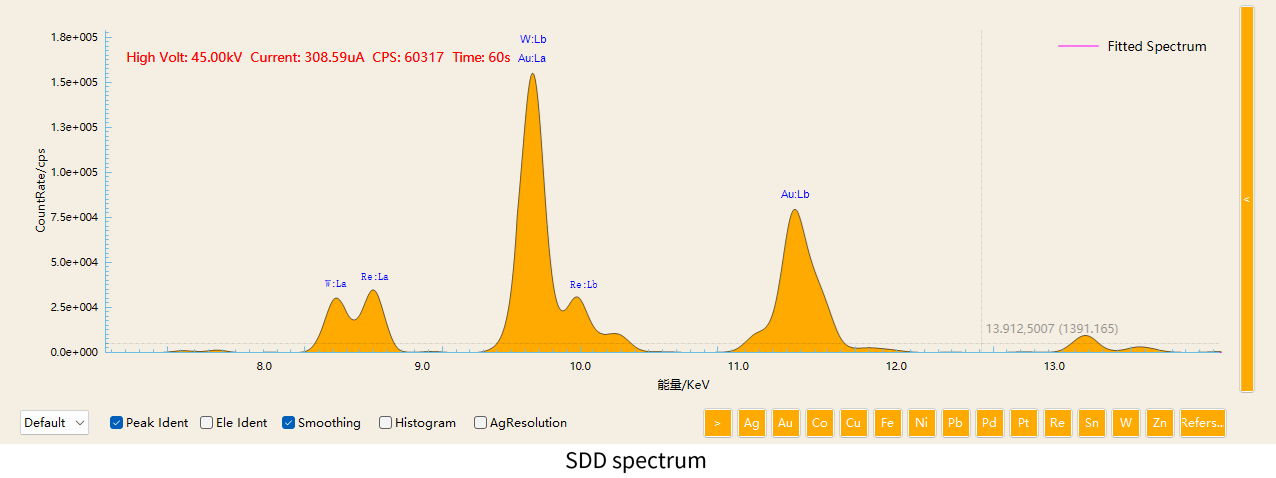
3. Separation of Au / Re / W Peaks
In some specialized material, Au, Re, and W may coexist. Their characteristic X-ray energies are extremely close:
The energy difference between W and Re is only about 260 eV. With low-resolution detectors, these peaks may significantly overlap.
However, high-resolution SDD detectors can clearly separate these peaks, greatly improving element identification accuracy.
4. Stability from High Count Rate
Precious metal analysis often requires rapid and stable measurement results.
SDD detectors support count rates above 500 kcps. With a 25 mm² detector area (vs 6 mm² for SIPIN), FSDD can achieve nearly three times the count rate under the same conditions.
More importantly, FSDD maintains good energy resolution at high count rates, whereas SIPIN detectors show significant degradation.
Higher count rates result in:
• Lower statistical error
• Shorter measurement time
• Better repeatability
This stability is especially important in jewelry retail testing and precious metal recycling.
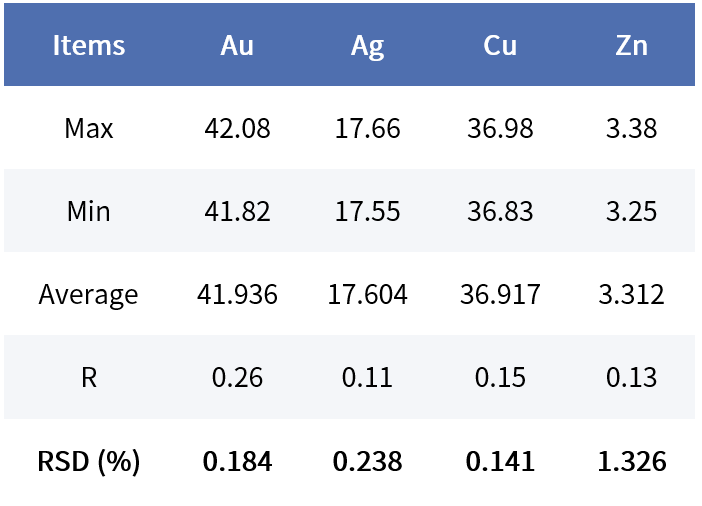
Taking K-gold testing stability as an example, 10 measurements of the same sample were compared using a SIPIN instrument (30,000 cps) and an FSDD instrument (80,000 cps).
Gold Lite:
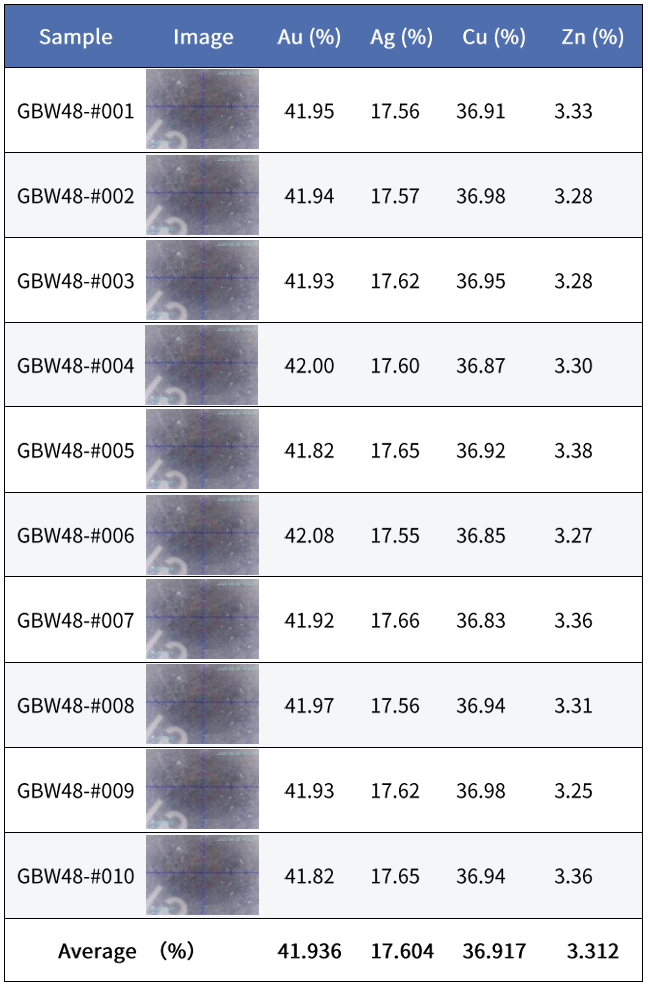
GoldSDD:
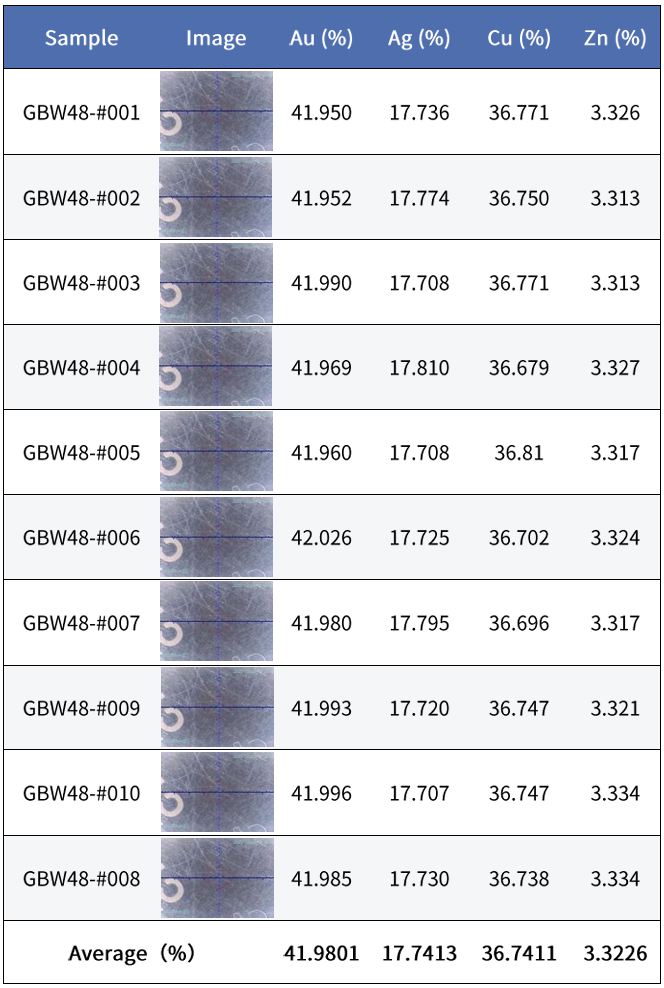
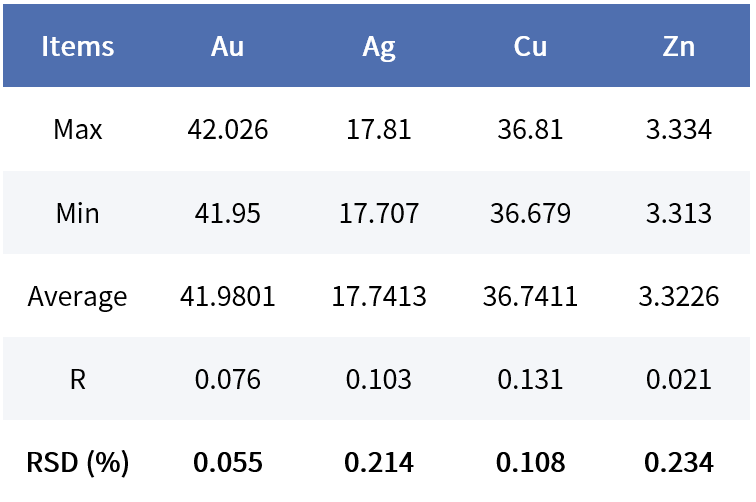
Considering customers’ demand for shorter testing times, under the same total counts and with comparable stability, the SIPIN instrument requires about 60 seconds, whereas the FSDD instrument needs only about 25 seconds.
5. Application Advantages
Due to its high resolution and high count-rate capability, the FSDD detector offers significant advantages in precious metal testing, including:
• Clearer elemental peak separation
• More stable analytical results
• Higher testing efficiency
For this reason, modern high-end precious metal XRF instruments increasingly adopt FSDD detectors.
-

-

-

-
 Probe into Novel Gold-plated Jewelry2025-06-04
Probe into Novel Gold-plated Jewelry2025-06-04 -
 XRF Secondary Programming - "Formula Editor"2025-03-23
XRF Secondary Programming - "Formula Editor"2025-03-23 -